Alpha particles6/25/2023 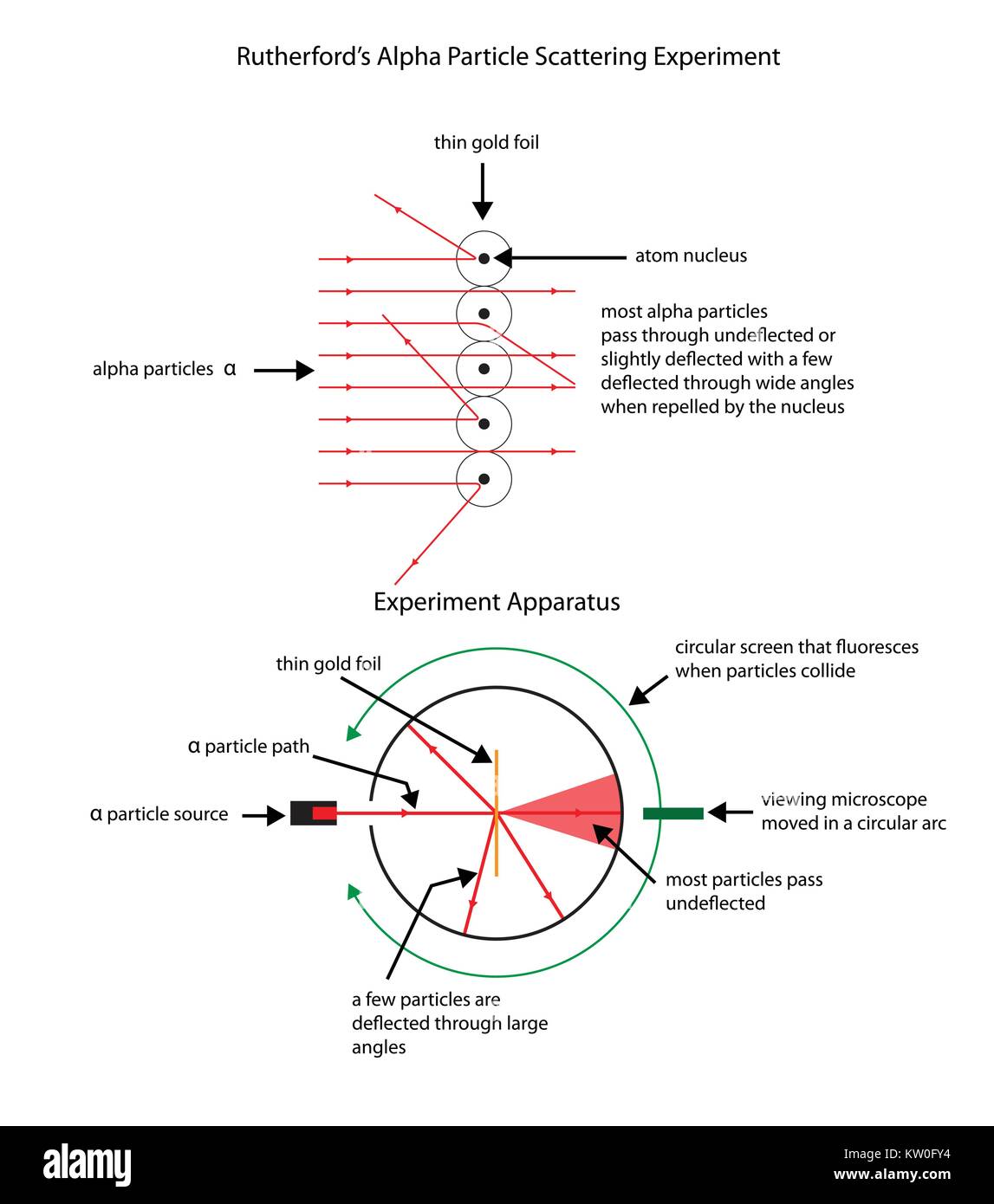
Both the negative and positive charges within the Thomson atom are spread out over the atom's entire volume, and Rutherford had calculated that this volume was too large for strong deflection to happen. Thomson's model predicted that the electric fields in an atom are too weak to affect a passing alpha particle much, given how fast and heavy alpha particles are. Thus the alpha particle will not bounce off the atom like a tennis ball hitting a basketball, but will pass right through if the atom's electric fields are weak enough to permit it. The Thomson atom is a sphere of electric charge anchored in space by its mass. At the atomic scale, the concept of "solid matter" is meaningless. Īccording to Thomson's model, if an alpha particle were to collide with an atom, it would just fly straight through, its path being deflected by at most a fraction of a degree. Rutherford discovered their existence and deduced that they were essentially helium atoms without electrons, though nobody knew about protons and neutrons at the time. Īn alpha particle is a sub-microscopic, positively charged particle of matter that is spontaneously emitted from certain radioactive elements. 
He proposed instead that electrons orbit the positive charge like the rings around Saturn. Japanese scientist Hantaro Nagaoka rejected Thomson's model on the grounds that opposing charges cannot penetrate each other. Thomson himself was never able to develop a complete and stable model of his concept. Thomson's model was not universally accepted even before Rutherford's experiments. This model was based entirely on classical (Newtonian) physics the current accepted model uses quantum mechanics. The existence of protons and neutrons was unknown at this time. Thomson had discovered the electron and believed that every atom was a sphere of positive charge throughout which the electrons were distributed, a bit like raisins in a Christmas pudding. The prevailing theory of atomic structure at the time of Rutherford's experiments was the " plum pudding model". 
The plum pudding model of the atom, as envisioned by Thomson
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
